
The most common sites of disease were the pancreas (37.4%) and small intestine (23.4%). The median age was 59.0 years (range, 29–80) and 44.9% had ECOG performance status (PS) 1. Baseline characteristics are shown in Table 1. Median duration of pembrolizumab treatment was 4.5 months (range, 0.03–25.3), and the median number of pembrolizumab doses was 7 (range, 1–35). 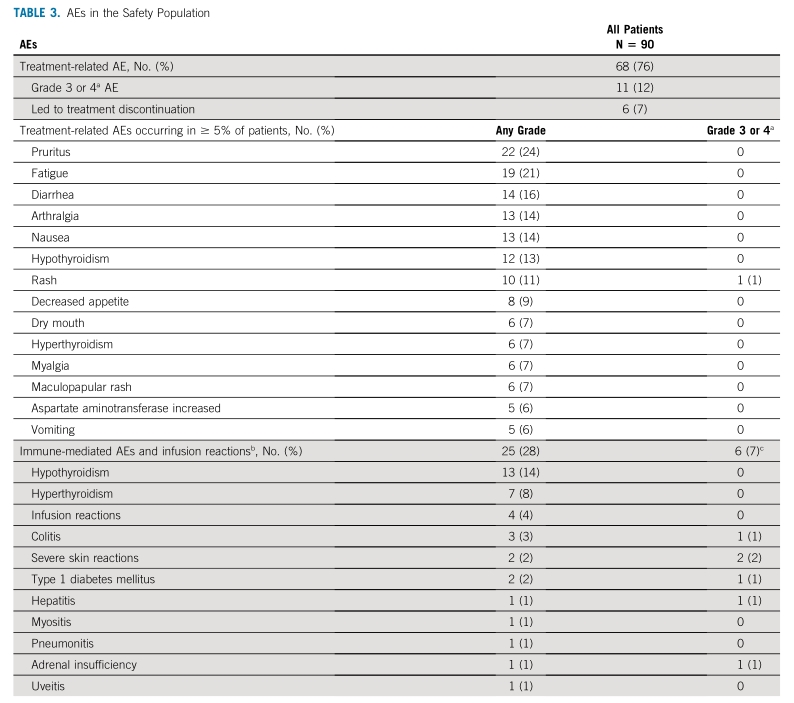
Overall, 104 (97.2%) patients discontinued pembrolizumab, most commonly for disease progression (Supplementary Fig. As of the December 6, 2018, data cutoff, the median follow-up duration was 24.2 months (range, 0.6–33.4). All patients had received ≥1 dose of pembrolizumab. Patients were enrolled regardless of tumor biomarker expression.įrom February 23, 2016, to August 03, 2016, 107 patients were enrolled at 42 sites in 16 countries (Supplementary Table S1). All patients were required to provide tumor tissue from a newly obtained core or excisional biopsy sample (preferred) or archival tumor sample of a nonirradiated lesion for PD-L1 assessment. Key eligibility criteria for the NET cohort included age ≥18 years well and moderately differentiated NET of the lung, appendix, small intestine, colon, rectum, or pancreas progression on or intolerance to ≥1 line of standard therapy measurable disease as assessed by Response Evaluation Criteria in Advanced Solid Tumors version 1.1 (RECIST v1.1) per independent central radiologic review Eastern Cooperative Oncology Group (ECOG) performance status of 0 or 1 and adequate organ function. 
In brief, KEYNOTE-158 is an international, open-label, phase II study of single-agent pembrolizumab across multiple advanced solid tumor types that have progressed on standard-of-care systemic therapy. The study design of the KEYNOTE-158 clinical trial ( identifier: NCT02628067) has been described previously ( 21). Here, we present the results from the cohort of biomarker unselected patients with previously treated advanced well-differentiated NETs enrolled in KEYNOTE-158. The KEYNOTE-158 phase II basket study investigated the antitumor activity and safety of pembrolizumab monotherapy in multiple cancer types. Moreover, durations of response were 6.9, 9.2, and 11.1 months for the carcinoid responders and the pNET responder had an ongoing response of 17.6 months ( 20). Overall, three patients with carcinoid (12% 95% CI, 3%–31%) and one patient with pNETs (6% 95% CI, 0%–30%) had objective responses, and SD rates were 60% ( n = 15) and 88% ( n = 14), respectively ( 20). Single-agent pembrolizumab showed antitumor activity in some patients with previously treated, PD-L1–positive carcinoid and pNETs in the phase Ib KEYNOTE-028 study ( 20). One such immune checkpoint inhibitor is pembrolizumab, a highly selective, humanized mAb that blocks the interaction of PD-1 with its ligands, PD-L1 and PD-L2 ( 19). Immune checkpoint inhibitors have demonstrated antitumor activity in many tumor types. Furthermore, associations between higher PD-L1 expression and decreased survival have been reported in metastatic GEP-NETs and pulmonary NETs ( 14, 18). Similar associations between PD-L1 expression and tumor grade have been reported in metastatic gastroenteropancreatic neuroendocrine tumors (GEP-NET ref. Consistent with these findings, expression of PD-L1 was rare among archival tissue samples from low-grade NETs of the small intestine ( n = 64) and pancreas ( n = 31 ref. Across NET sites, PD-L1 expression was detected in 0% of grade 1, 78% of grade 2, and 100% of grade 3 tumors ( 16). For example, PD-L1 expression was reported in 59% of pulmonary NETs ( 14) and 54% of insulinoma-like pancreatic NETs (pNET ref. Several studies suggest that programmed death ligand 1 (PD-L1) expression plays a role in the development, progression, and prognosis of NETs, especially in high-grade tumors. In cancer, PD-1 promotes tumor escape from host immune responses ( 12, 13). Programmed death 1 (PD-1) is a T-cell coinhibitory receptor that regulates immune response by interacting with its ligands (PD-L) ( 11).
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
